Adult T-cell leukemia/lymphoma (ATLL) is a rare and aggressive disease of malignant CD4+ T cells that develops in human T-lymphotropic virus-1 (HTLV-1) carriers. Patients diagnosed with ATLL have the worst survival among all peripheral T-cell lymphomas. There is an urgent need for in-depth understanding of its pathogenesis and development of new treatment strategies. We and others have shown that ATLLs diagnosed in the Japanese (J-ATLL) and North American (NA-ATLL) patients have differences in clinical characteristics and immune-genetic landscape. Among the most frequently mutated genes in J-ATLL cohorts is CCR4, a chemokine receptor that is targeted by C-terminal truncation mutations in one third of the cases. These mutations result in a more stabilized surface CCR4 and have been reported to cause elevated Akt activation following chemokine stimulation. Since our previous targeted exon sequencing study did not include CCR4, this work was initiated to assess CCR4 mutation frequency in NA-ATLL cases and to evaluate the preclinical activity of a small molecule CCR4 antagonist. This is because, although an anti-CCR4 mAb, mogamulizumab, has been approved in Japan to treat ATLL, this therapy failed in a Phase 2 trial among patients outside of Japan. Thus, mogamulizumab is not available as a routine treatment for NA-ATLL patients.
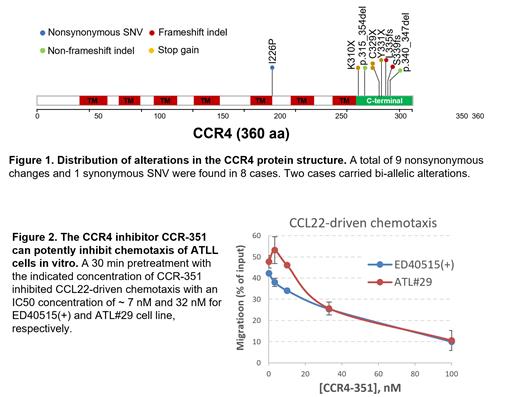
We compiled CCR4 gene status data from bulk RNA-seq (n=10) and whole genome sequencing (WGS) analysis (n = 10). Out of a total of 17 NA-ATLL cases, 8 (47.1%) carried CCR4 gene alterations that included in-frame and frameshift indels, as well as nonsense mutations targeted to the C-terminus cytoplasmic tail and also the trans-membrane domain. In subtype analysis, 6 out 14 acute cases (42.9%) and 2 out of 3 (66.7%) of chronic cases carried at least one copy of mutated CCR4 gene, indicating that the mutation frequency in NA-ATLL is possibly even higher than that reported for the Japanese cohorts ( Figure 1). We then evaluated cell surface CCR4 expression using flow cytometry. The primary ATLL cells (CD4 +CD7 -CD8 -) from all 7 diagnostic samples uniformly express markedly increased levels of CCR4 relative to healthy control CD4 T cells. Interestingly, CCR4 expression among 8 ATLL cell lines is highly variable, implying that in vivo, tumor microenvironment factors may also contribute to sustained CCR4 upregulation in addition to the influence from the HTLV-1 oncoprotein, HBZ.
FLX475 is an orally active small molecule CCR4 antagonist that is currently in a Phase 1/2 study (NCT03674567) in several types of advanced cancer including EBV+ NK/T cell lymphoma. So far, favorable clinical antitumor activity has been observed that includes complete responses with FLX475 monotherapy and encouraging combination activity (Lin C.-C. et al ESMO-IO 2022 Dec; #187P). Using several CCR4-positive cell lines, we examined in vitro anti-ATLL activity of CCR4-351, a CCR4 antagonist that is a highly related analog of FLX475. CCR4 is the exclusive homing receptor for two chemokine molecules, CCL17 and CCL22. In a transwell assay, both chemokines can elicit strong chemotaxis activity in a J-ATLL cell line ED40515(+). However, only CCL22 but not CCL17 can induce significant chemotaxis in two NA-ATLL cell lines, ATL#13 and ATL#29. The exact cause of this difference between these cell lines awaits future studies. In these transwell-based assays, CCR4-351 exhibited potent chemotaxis inhibitory activity with IC 50 values ranging from 7-60 nM ( Figure 2). In addition, when the cell lines were cultured under growth factor-deprivation conditions, CCR4-351 treatment caused a mild reduction in total cell viability, suggesting an additional anti-ATLL mechanism of action.
In summary, in this first study of the CCR4 pathway status in NA-ATLL, we found that nearly half of the cases in our ATLL cohort carried a mutated CCR4 gene. Contrary to primary ATLL cells, ATLL cell lines exhibit a wide variation in CCR4 expression. CCL22 but not CCL17 induced strong chemotaxis behavior in NA-ATLL cell lines, which was potently inhibited by a small molecule CCR4 antagonist, CCR4-351. Since extramedullary presentation is frequently seen in NA-ATLL and central nervous system involvement is an adverse prognostic feature, inhibiting chemotaxis with a CCR4 antagonist such as FLX475 may be an effective therapeutic approach.
Disclosures
Ye:Rapt Therapeutics, Inc: Research Funding. Shah:Plantable: Research Funding; Janssen: Consultancy, Other: Advisory Board, Research Funding; Sanofi: Other: Advisory Board; C4 Therapeutics: Research Funding; Sabinsa: Research Funding; M and M Labs: Research Funding; Bristol Myers Squibb: Consultancy, Other: Advisory Board, Research Funding. Shastri:Rigel Pharmaceuticals: Honoraria; Kymera Therapeutics: Honoraria, Research Funding; Gilead Sciences: Honoraria; Janssen Pharmaceuticals, Inc.: Consultancy, Honoraria. Kassner:Rapt Therapeutics, Inc: Current Employment, Current equity holder in publicly-traded company. Verma:Prelude: Research Funding; Curis: Research Funding; Medpacto: Research Funding; Eli Lilly: Research Funding; Incyte: Research Funding; GSK: Research Funding; BMS: Research Funding; Throws Exception: Current equity holder in private company; Janssen: Honoraria; Celgene: Other: Scientific Advisor; Acceleron: Other: Scientific Advisor; Novartis: Other: Scientific Advisor; Bakx: Current equity holder in private company, Other: Scientific Advisor; Stelexis: Current equity holder in private company, Honoraria, Other: Scientific Advisor.